 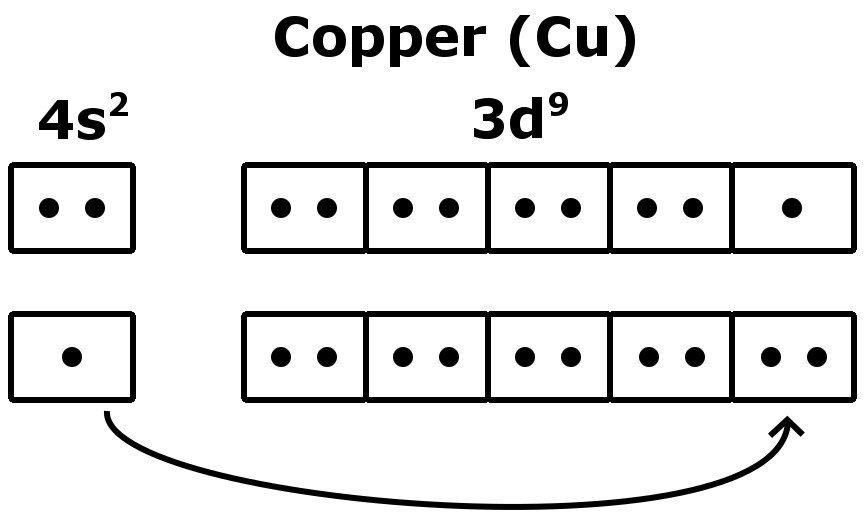 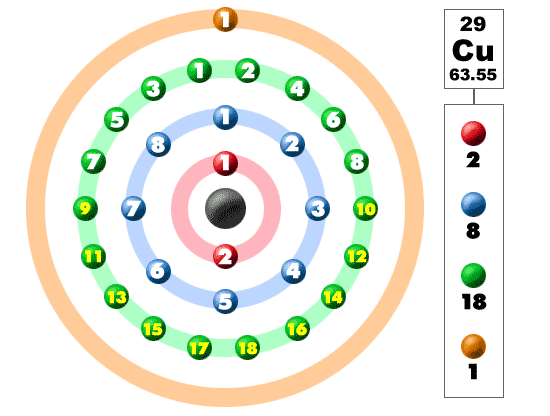
This is due to the fact that the atoms and electrons know nothing of the rules we (humans) come up with to try to explain and predict their behavior. 
For example, W will follow the Aubau principle and not display behavior like that of Cr. 
There are other elements which will have exceptions to the rules we have come up with to predict electron configurations. Unfortunately, there is no easy way to explain these deviations in the ideal order for each element. Interestingly enough, Tungsten is more stable with an electron arrangement of Xe4f 145d46s2. It is important to note that this explanation is just looking at the examples of Cr and Cu. The electron configuration for chromium is NOT 1s22s22p63s23p63d44s2, but 1s22s22p63s23p63d54s1. Members of a group typically have similar properties and electron configurations in their. This gives the atom greater stability so the change is favorable. Element Copper (Cu), Group 11, Atomic Number 29, d-block, Mass 63.546. In both examples, an electron moves from the 4s sublevel to produce a 1/2 full 3d (Cr) or completely filled 3d (Cu). Electrons are lazy and will do whatever places them in the lowest energy state = which is the most stable state A sublevel which is exactly half filled is more stable than a partially filled sublevel which is not half full.ģ. Completely filled sublevels are more stable than partially filled sublevels.Ģ. To understand why this occurs, it is important to realize that.ġ. The electronic configuration of Copper will be 1s2 2s2 2p6 3s2 3p6 3d10 4s1.Using the Aufbau principle, you would write the following electron configurations How do you write the electron configuration for Copper? The electronic configuration of Copper will be 1s2 2s2 2p6 3s2 3p6 3d10 4s1. What is the electronic configuration of Copper 29? What is the boiling Point of Copper in Kelvin?īoiling Point of Copper in Kelvin is 3200 K. Melting Point of Copper in Kelvin is 1357.77 K. What is the melting Point of Copper in Kelvin? What is the boiling Point of Copper?īoiling Point of Copper is 3200 K. Copper has 29 electrons out of which 2 valence electrons are present in the 3d10 4s1 outer orbitals of atom. How many valence electrons does a Copper atom have?Ĭopper has 2 valence electrons. Copper was first isolated by Anatolia in 6000 BCE. The element Copper was discovered by Middle East in year 9000 BCE. What is the color of Copper?Ĭopper is of Copper color. It is located in group 11 and period 4 in the modern periodic table. Copper is the 29 element on the periodic table. What is the position of Copper in the Periodic Table?Ĭopper is a chemical element with the symbol Cu and atomic number 29. Copper is a chemical element with symbol Cu and atomic number 29. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Copper is 3d10 4s1. What is the abbreviated electronic configuration of Copper? The electronic configuration of Copper is 1s2 2s2 2p6 3s2 3p6 3d10 4s1. What is the electronic configuration of Copper? Optical Properties of Copper Refractive IndexĪcoustic Properties of Copper Speed of SoundĬopper Thermal Properties - Enthalpies and thermodynamics Refer to table below for the Electrical properties ofCopper Electrical ConductivityĬopper Heat and Conduction Properties Thermal Conductivity Hardness of Copper - Tests to Measure of Hardness of Element Mohs HardnessĬopper is Conductor of electricity. Refer to below table for Copper Physical Properties DensityĨ.92 g/cm3(when liquid at m.p density is $8.02 g/cm3) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
